
Cleanroom testing, certification, and environmental validation in Singapore
Independent third-party testing to ISO 14644, NEBB, and GMP standards. Covering cleanrooms, laminar flow cabinets, HVAC systems, and controlled environments.
Overview
Clean Aire delivers Singapore's most comprehensive third-party cleanroom testing suite — certification, HVAC airflow balancing, microbiological monitoring, smoke studies, and controlled-environment validation to ISO 14644 and NEBB standards.
Every measurement is performed by NEBB-certified technicians and backed by ISO 9001:2015, SAC accreditation, and bizSafe3 — findings that stand up to pharmaceutical, semiconductor, healthcare, and research audits without compromise.
-
 NEBB Certified National Environmental Balancing Bureau
NEBB Certified National Environmental Balancing Bureau -
 ISO 9001:2015 Quality Management System Certified
ISO 9001:2015 Quality Management System Certified -
 SAC Accredited Singapore Accreditation Council certification body
SAC Accredited Singapore Accreditation Council certification body -
 bizSafe3 Workplace Safety & Health
bizSafe3 Workplace Safety & Health

Services
From third-party cleanroom certification to HVAC airflow balancing, microbiological monitoring, and airflow visualisation, we deliver a full suite of controlled-environment testing to ISO 14644 and NEBB standards. Each service is calibrated to the regulatory, safety, and operational demands of Singapore's pharmaceutical, semiconductor, and healthcare facilities.
- 01
Cleanroom Certification & Performance Testing
Independent third-party ISO 14644 and NEBB certification — particle counts, airflow visualisation, air-change rates, recovery tests, and temperature and humidity mapping across your controlled environment.
- 02
Clean Air Equipment Testing
Performance validation for laminar flow cabinets, clean booths, clean air devices, fume hoods, biological safety cabinets, and isolators — protecting both product and operator.
- 03
HVAC Airflow Measurement & Balancing
Precise airflow measurement and balancing to meet pressure cascade, room change-rate, and return-air targets across critical controlled environments.
- 04
Microbiological & Air Quality Monitoring
Viable and non-viable particulate sampling, environmental microbial monitoring, and indoor air quality assessments for audit-ready hygiene evidence.
- 05
Compressed Dry Air Testing
Quality and purity testing of compressed dry air and process gas systems feeding cleanroom production — dewpoint, oil, and particulate verification to ISO 8573.


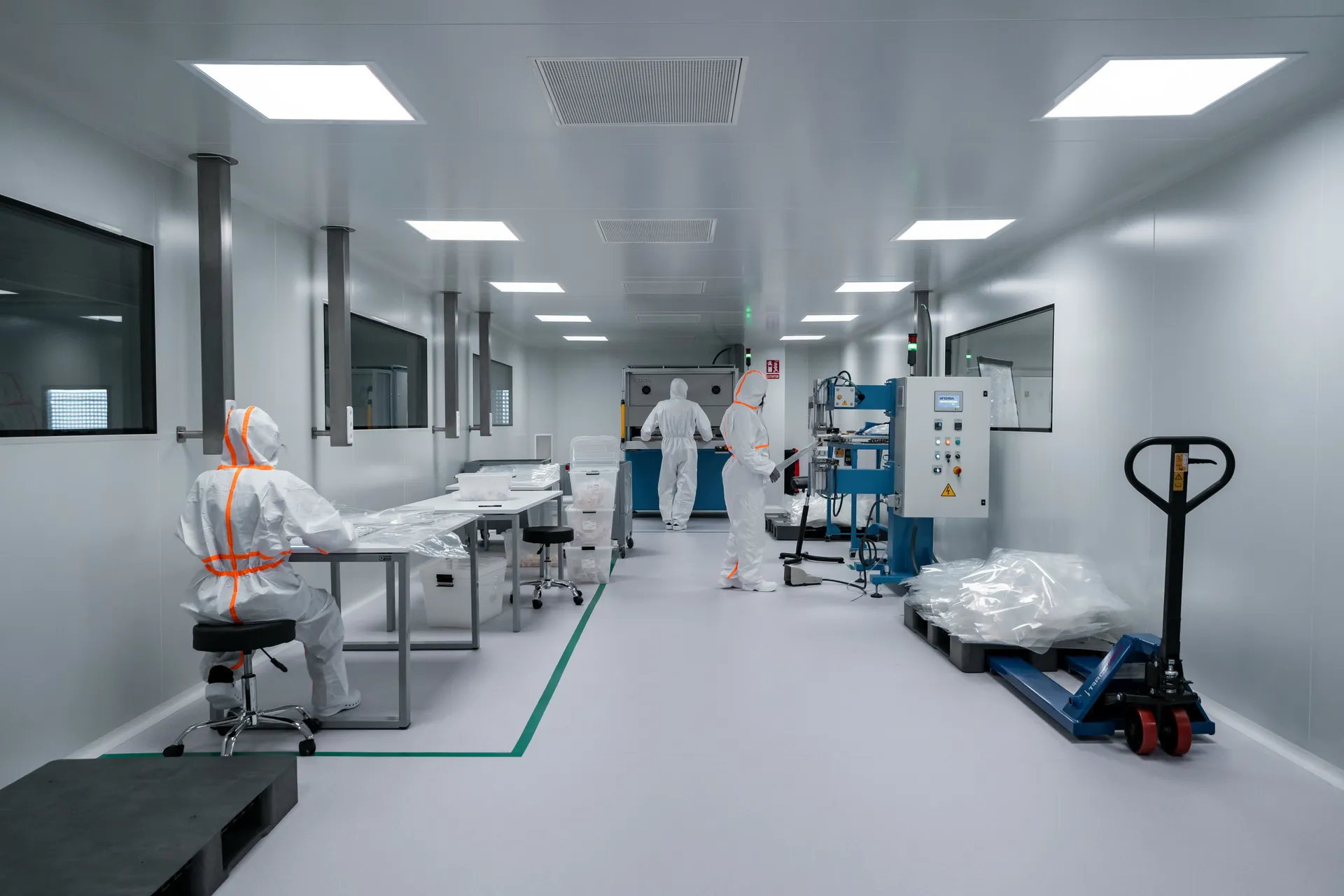


Our testing and certification services support pharmaceutical, semiconductor, healthcare, and research facilities across Singapore and the region.
- Pharmaceuticals
- Semiconductors / Electronics
- Healthcare
- Food & Beverages
- Bio-Science
- Schools & Institutions
- Research Laboratories
- Pfizer
- Novartis
- Roche
- AbbVie
- BioNTech
- Mead Johnson
- STMicroelectronics
- UMC
- Soitec
- Coherent
- National University Hospital
- DSO National Laboratories
Ready to put our test methods to work in your facility?
From initial third-party certification to annual re-qualification — our NEBB-certified team delivers the accredited, audit-defensible results your regulators, clients, and insurers demand.