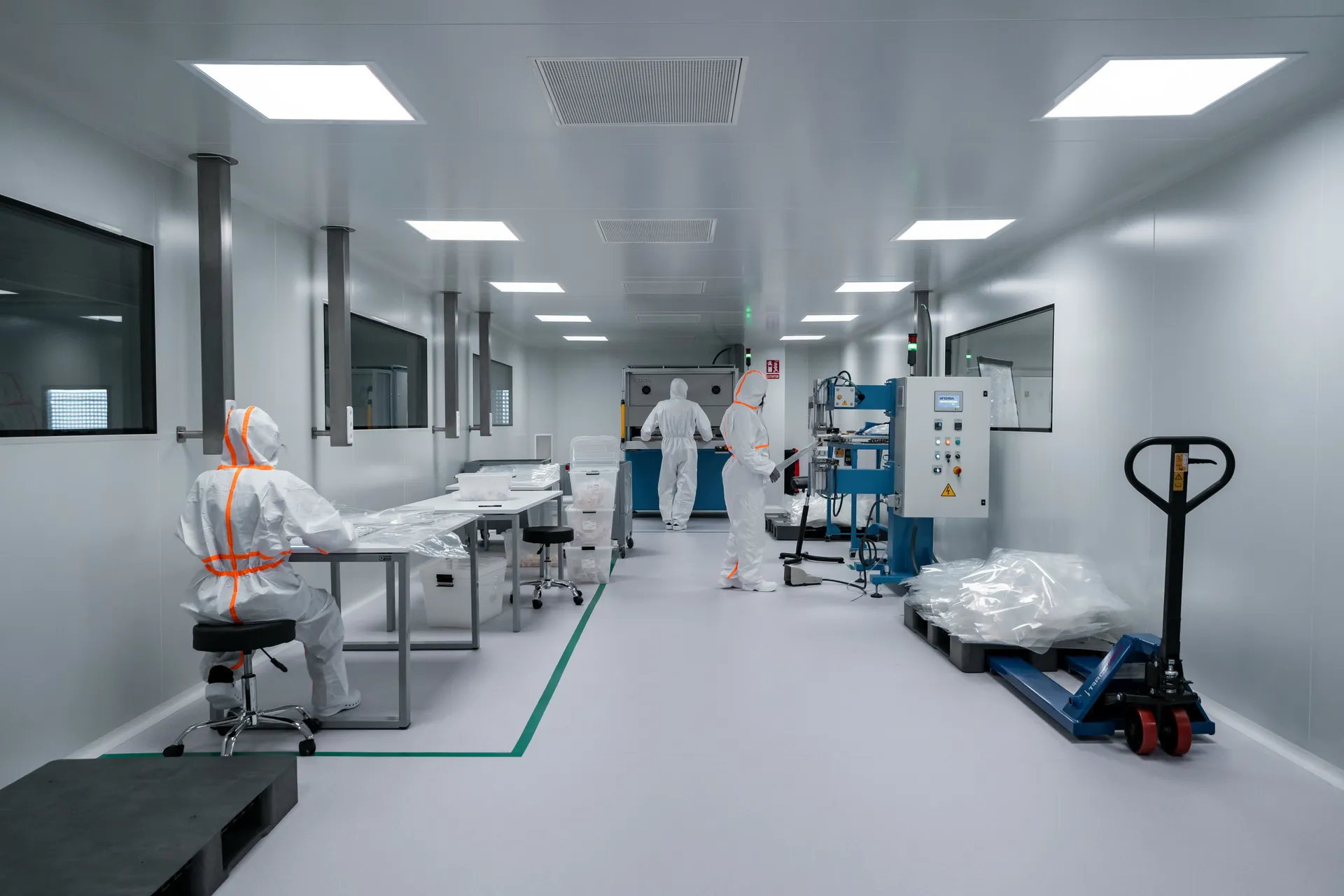
Cleanroom performance testing and ISO 14644 certification in Singapore
Independent, NEBB-certified testing to ISO 14644, FS 209E, and IEST standards. Covering cleanrooms, laminar flow cabinets, HVAC systems, controlled environments, and compressed gases.
Overview
Clean Aire is an independent, NEBB-certified cleanroom testing and certification specialist delivering ISO 14644-compliant measurement across pharmaceutical, semiconductor, healthcare, and research facilities since 2010.
Our ISO 9001:2015-governed methodology combines on-site testing to internationally recognised standards (ISO 14644, FS 209E, IEST RP), traceable instrumentation calibration, and peer-reviewed reports — evidence your regulators, auditors, and insurers accept without question.
-
 NEBB Certified National Environmental Balancing Bureau
NEBB Certified National Environmental Balancing Bureau -
 ISO 9001:2015 Quality Management System Certified
ISO 9001:2015 Quality Management System Certified -
 SAC Accredited Singapore Accreditation Council certification body
SAC Accredited Singapore Accreditation Council certification body -
 bizSafe3 Workplace Safety & Health
bizSafe3 Workplace Safety & Health

Services
From third-party cleanroom certification to HVAC airflow balancing, microbiological monitoring, and airflow visualisation, we deliver a full suite of controlled-environment testing to ISO 14644 and NEBB standards. Each service is calibrated to the regulatory, safety, and operational demands of Singapore's pharmaceutical, semiconductor, and healthcare facilities.
- 01
Cleanroom Certification & Performance Testing
ISO 14644-1 classification, ISO 14644-2 monitoring, and ISO 14644-3 test methods — non-viable particle counts, recovery tests, pressure cascades, and HEPA installation leak tests, delivered as peer-reviewed certification reports.
- 02
Clean Air Equipment Testing
Face velocity, HEPA integrity via PAO/DOP aerosol challenge (IEST-RP-CC034), containment testing per NSF/ANSI 49, and smoke-pattern visualisation — covering laminar flow cabinets, clean booths, biological safety cabinets, fume hoods, and isolators.
- 03
HVAC Airflow Measurement & Balancing
NEBB-certified procedures for air-change-rate calculation, pressure-differential verification, and multi-point Pitot-traverse balancing — ensuring supply, return, and exhaust conform to facility design intent and GMP operational envelopes.
- 04
Microbiological & Air Quality Monitoring
Viable sampling via settle plates and active air impaction (TSA/SDA), contact-plate swabbing, non-viable particulate counts per ISO 14644-1, and SS554 indoor air quality — CO₂, CO, formaldehyde, VOCs, and particulates for pharmaceutical, healthcare, and educational environments.
- 05
Compressed Dry Air & Process Gas Testing
Dewpoint, oil aerosol, oil vapour, particulate, and microbiological verification to ISO 8573-1 purity classes — validating the compressed air, nitrogen, and process gases feeding your critical production environments.





Our testing and certification services support pharmaceutical, semiconductor, healthcare, and research facilities across Singapore and the region.
- Pharmaceuticals
- Semiconductors / Electronics
- Healthcare
- Food & Beverages
- Bio-Science
- Schools & Institutions
- Research Laboratories
- Pfizer
- Novartis
- Roche
- AbbVie
- BioNTech
- Mead Johnson
- STMicroelectronics
- UMC
- Soitec
- Coherent
- National University Hospital
- DSO National Laboratories
Need your cleanroom tested to ISO 14644 or FS 209E?
Our NEBB-certified team delivers audit-ready particle counts, recovery tests, smoke studies, and HVAC balancing — with peer-reviewed reports regulators and GMP auditors accept without question.